High-Pressure Cylinders
and Green Energy
Why Bobson Holds DOT 3AL, TÜV, and ISO 13485 Certifications at the Same Time
In high-pressure vessels and medical-related supply chains, the word certification is often used as a generic term. In reality, DOT, TÜV, and ISO represent three fundamentally different systems, each addressing a different level of risk and responsibility.
Without clearly distinguishing between them, it is easy to assume that one certification is enough—or to mistake them as interchangeable labels. In high-risk manufacturing industries, however, companies that are genuinely trusted by global markets rarely rely on a single system.
Before discussing Bobson, it is essential to first understand what each of these frameworks truly represents.

| Category | DOT (DOT 3AL as example) | TÜV | ISO (ISO 13485 as example) |
|---|---|---|---|
| System Origin | Government regulation | Third-party verification system | International standard |
| Representative Body | United States Department of Transportation | TÜV Rheinland | International Organization for Standardization |
| Nature | Mandatory (non-compliance = illegal) | Independent third-party verification | Globally accepted management system standard |
| Management Level | Product / pressure vessel specification | Verification role | Company-wide quality management system |
| Core Question | Is it legal and safe to use? | Has compliance been independently verified? | Can risks be controlled consistently over time? |
| Material Control | ✔ | Depends on scope | ✔ |
| Process Control | ✔ (product-specific) | Depends on audit | ✔✔✔ |
| System Control | ✖ | Indirect | ✔✔✔ |
| Third-Party Audit | ✔ | ✔✔✔ | ✔ |
| One-Line Summary | Can it be used legally? | Is there independent proof? | Can the entire system sustain risk long-term? |
The key takeaway from this table is simple: DOT, TÜV, and ISO solve three different layers of risk—and none of them replaces the others.
How Government Regulations Define the Safety Baseline
DOT originates from the United States Department of Transportation and is part of a government regulatory framework. Its purpose is not to enhance quality—it establishes the minimum acceptable level of public safety.
In the field of pressure vessels, DOT is not a single certificate but a collection of legally defined specifications. DOT 3AL specifically governs seamless aluminum high-pressure cylinders.
The DOT framework addresses a straightforward question: Does this pressure vessel pose an unacceptable risk to people or public safety during transportation and use?
To answer that, DOT 3AL regulates:
If a cylinder does not comply, it simply cannot be legally transported or used.
For manufacturers, the real value of DOT 3AL is not passing a single test. Instead, it forces manufacturing processes to be verifiable, traceable, and repeatable.
This is why DOT compliance is typically the starting point for high-pressure vessel manufacturing—not the finish line.
How Third-Party Verification Builds Market Trust?
TÜV (Technischer Überwachungsverein) is neither a law nor a standard. It is a third-party technical verification system that originated in Germany. Organizations such as TÜV Rheinland play a specific role: To stand independently between manufacturers and the market, conducting audits and testing based on defined standards.
In international supply chains, buyers, regulators, and compliance teams face a common challenge: How can we quickly assess whether a manufacturer is trustworthy?
TÜV helps answer that question by:
TÜV does not guarantee quality outcomes. It confirms only one thing: Whether the organization truly meets the standards it claims to follow. For high-risk industries, this distinction is critical. TÜV is a trust mechanism, not a marketing badge.
International Consensus on Management System Capability
ISO standards are issued by the International Organization for Standardization. While ISO standards are not laws, they have become the shared language of global supply chains. ISO does not evaluate individual products. Instead, it asks: Does the organization have a management system capable of delivering consistent, auditable results over time?
ISO 13485 is a quality management system standard designed specifically for medical devices and their supply chains. Its requirements are significantly stricter than general manufacturing standards such as ISO 9001.
Key areas of focus include:
The objective is simple: Even as production scales, personnel changes, or product lines expand, quality must remain under control.
Once the roles of DOT, TÜV, and ISO are clearly understood, the real question emerges: If meeting the minimum legal requirement is enough to ship products, why invest further in third-party verification and medical-grade quality systems?
For Bobson, the answer is straightforward: Risk does not exist at only one level.
Together, these frameworks form a risk management architecture that global markets can understand, audit, and trust—not merely a collection of certificates.
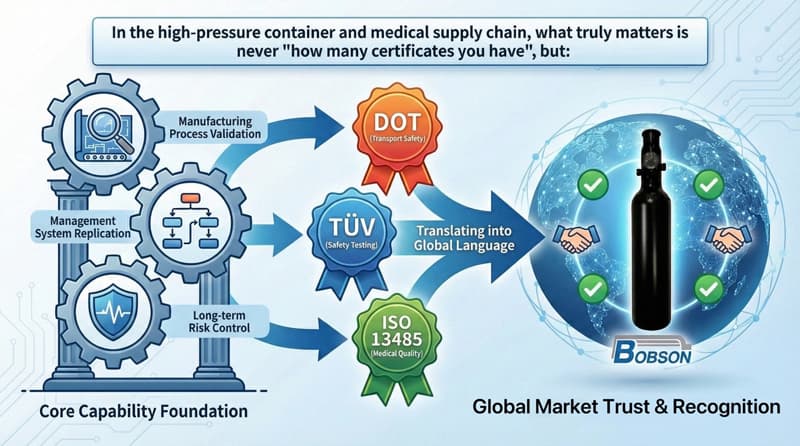
In high-pressure vessel and medical supply chains, the real question is never how many certificates a company holds. The real questions are:
DOT, TÜV, and ISO 13485 translate those capabilities into a language that global markets recognize. That is why Bobson chooses to implement all three—not as labels, but as a foundation for long-term trust.